Background Information
Humulone, being light sensitive, degrades in light. This degradation is minimized by keeping reactions out of light and bottling the beverage in colored glass bottles. This adds more cost to the manufacture process as colored glass is more costly than clear glass bottles. If the isomerization of humulone can be manipulated, the price of manufacturing beer will drop, in turn allowing consumers a price drop as well (Keukeleire, D. D., 2000).
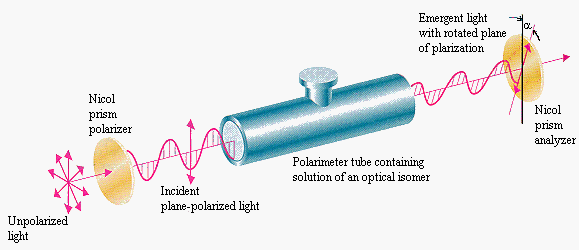
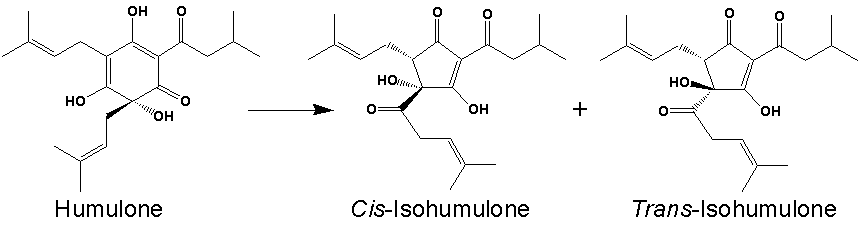
Humulone rotates polarized light at -212° per g/mL. This is humulone’s optical rotation. Cis- and trans- isohumulone have an optical rotation of 46.7° and -7.8° respectively. Knowing this, we can calculate the amounts of cis- and trans- isohumulone using a polarimeter during and after isomerization.
Sharpe and Ormrod used a preparation of an o-phenylenediamine/ humulone complex from 150 g of an alpha-acids-rich hop extract and 600 mL of benzene. The mixture they used was stirred on a hotplate with 12 g of decolorizing charcoal and 55 g o-phenylenediamine added prior to the mixture boiling. Sharpe and Ormrod refluxed the mixture for two minutes and then added another 2 g of decolorizing charcoal before immediately filtering under vacuum through a pre-heated sintered glass funnel. The filtrate was then cooled to 5 degrees C and the precipitate was removed by the vacuum filtration. This was done another four times with decolorizing charcoal added to the first two times. This method of re-crystallization produced 90 g of complex.
Ensuing the re-crystallization came separation of humulone from the complex. This was achieved through a separatory funnel. 60 ml of 6 M hydrochloric acid was poured into a 250 ml glass separating funnel, and aluminum foil wrapped round the outer surface to exclude light. 5 g of the o-phenylenediamine/ humulone complex was added to the separating funnel, followed by 30 ml of ether. The funnel was stoppered, shaken vigorously, and vented frequently. The lower aqueous layer was slowly drained, with the organic layer being collected. The aqueous layer was re-extracted twice with two 30 ml aliquots of ether, and the organic layers collected and combined with the initial organic fraction in a second, light-shielded 250 ml separating funnel.
The combined ether extracts were then washed with 6 M HCl (50 ml), saturated NaCl (50 ml), and two 100 ml aliquots of distilled water. The mixture was allowed to separate, the aqueous layer discarded, and the ether evaporated by rotary evaporation. The remaining white solid was humulone. Using this procedure, 5 g of o-phenylenediamine complex yielded 3.8 g of humulone (Sharpe and Ormrod, 1991).
Francis L. Rigby found a way to get a concentrate of isohumulones. 3000 mL of hot water was added with 90 mL of 3.0 N NaOH and 80 g of hops and refluxed and boiled for 10 minutes. After boiling, the mixture was filtered to get the spent hops separated from the liquid. Then 74 mL of 6 N HCl was added and the acid formed a precipitate. The isohumulones adhere to this precipitate for extraction.
Following the patent is useful because getting an H-NMR spectrum of isohumulone will determine if using ammonium hydroxide is a faster way to get humulone to isomerize. H-NMR spectra will be used to compare the two humulone samples. If the ammonium hydroxide buffer works, it would be much easier to get humulone to isomerize after extraction from hops oil.
Humulone rotates polarized light at -212° per g/mL. This is humulone’s optical rotation. Cis- and trans- isohumulone have an optical rotation of 46.7° and -7.8° respectively. Knowing this, we can calculate the amounts of cis- and trans- isohumulone using a polarimeter during and after isomerization.
Sharpe and Ormrod used a preparation of an o-phenylenediamine/ humulone complex from 150 g of an alpha-acids-rich hop extract and 600 mL of benzene. The mixture they used was stirred on a hotplate with 12 g of decolorizing charcoal and 55 g o-phenylenediamine added prior to the mixture boiling. Sharpe and Ormrod refluxed the mixture for two minutes and then added another 2 g of decolorizing charcoal before immediately filtering under vacuum through a pre-heated sintered glass funnel. The filtrate was then cooled to 5 degrees C and the precipitate was removed by the vacuum filtration. This was done another four times with decolorizing charcoal added to the first two times. This method of re-crystallization produced 90 g of complex.
Ensuing the re-crystallization came separation of humulone from the complex. This was achieved through a separatory funnel. 60 ml of 6 M hydrochloric acid was poured into a 250 ml glass separating funnel, and aluminum foil wrapped round the outer surface to exclude light. 5 g of the o-phenylenediamine/ humulone complex was added to the separating funnel, followed by 30 ml of ether. The funnel was stoppered, shaken vigorously, and vented frequently. The lower aqueous layer was slowly drained, with the organic layer being collected. The aqueous layer was re-extracted twice with two 30 ml aliquots of ether, and the organic layers collected and combined with the initial organic fraction in a second, light-shielded 250 ml separating funnel.
The combined ether extracts were then washed with 6 M HCl (50 ml), saturated NaCl (50 ml), and two 100 ml aliquots of distilled water. The mixture was allowed to separate, the aqueous layer discarded, and the ether evaporated by rotary evaporation. The remaining white solid was humulone. Using this procedure, 5 g of o-phenylenediamine complex yielded 3.8 g of humulone (Sharpe and Ormrod, 1991).
Francis L. Rigby found a way to get a concentrate of isohumulones. 3000 mL of hot water was added with 90 mL of 3.0 N NaOH and 80 g of hops and refluxed and boiled for 10 minutes. After boiling, the mixture was filtered to get the spent hops separated from the liquid. Then 74 mL of 6 N HCl was added and the acid formed a precipitate. The isohumulones adhere to this precipitate for extraction.
Following the patent is useful because getting an H-NMR spectrum of isohumulone will determine if using ammonium hydroxide is a faster way to get humulone to isomerize. H-NMR spectra will be used to compare the two humulone samples. If the ammonium hydroxide buffer works, it would be much easier to get humulone to isomerize after extraction from hops oil.