Results
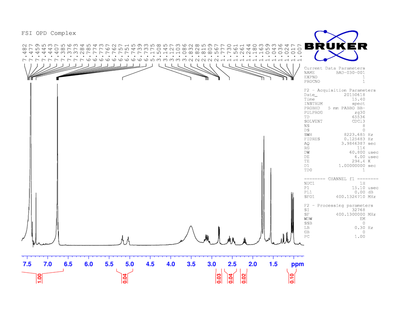
After making the o-phenylenediamine/ humulone complex, an H-NMR was taken to verify that the complex obtained was the complex desired, shown in Figure 1. The H-NMR does show that the desired complex was obtained. This is great because 34.472 g of the crystals were obtained.
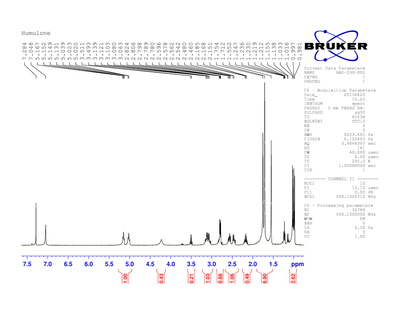
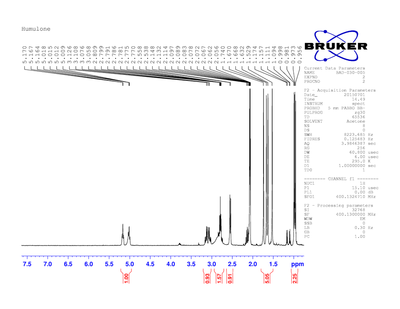
After the isolation of humulone, H-NMR spectra were obtained in two different deuterated solvents. The two solvents were used since each solvent has its own peak that is seen in the spectrum and the peak may overlap a peak present in the compound. Figure 2 shows humulone in deuterated chloroform and Figure 3 shows humulone in deuterated acetone. Figure 2 shows chloroform and benzene remnants in the sample. The chloroform and benzene peaks are the peaks to the far left in the spectrum. The benzene had just not evaporated fully and the chloroform is present because not all of the chloroform in the solvent was deuterated. The peak is not deuterated chloroform, it is just chloroform.
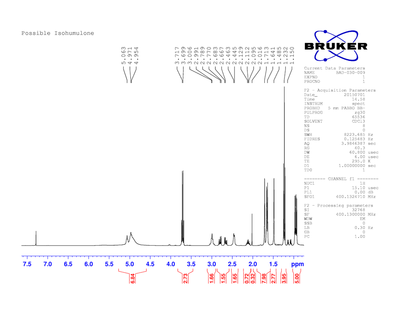
Possible isohumulone was used for an H-NMR spectrum. Figure 4 shows the spectrum of humulone after being dissolved in a basic solution of sodium hydroxide (NaOH). The humulone did not fully dissolve in the NaOH. This likely gave us skewed polarimeter results that are inconclusive.
After the isolation of humulone, H-NMR spectra were obtained in two different deuterated solvents. The two solvents were used since each solvent has its own peak that is seen in the spectrum and the peak may overlap a peak present in the compound. Figure 2 shows humulone in deuterated chloroform and Figure 3 shows humulone in deuterated acetone. Figure 2 shows chloroform and benzene remnants in the sample. The chloroform and benzene peaks are the peaks to the far left in the spectrum. The benzene had just not evaporated fully and the chloroform is present because not all of the chloroform in the solvent was deuterated. The peak is not deuterated chloroform, it is just chloroform.
Possible isohumulone was used for an H-NMR spectrum. Figure 4 shows the spectrum of humulone after being dissolved in a basic solution of sodium hydroxide (NaOH). The humulone did not fully dissolve in the NaOH. This likely gave us skewed polarimeter results that are inconclusive.
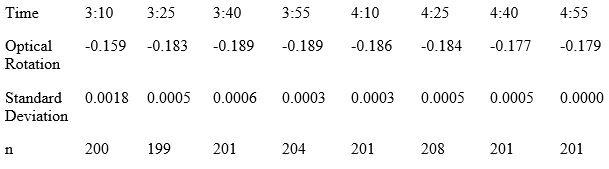
A polarimeter reading was taken for humulone that was dissolved in DI water with a pH of 10 (Below).
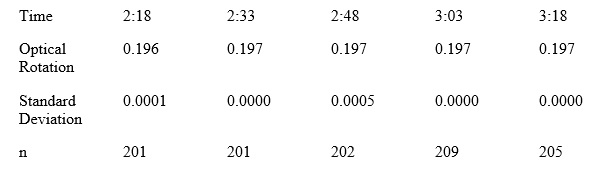
A polarimeter reading was taken of the humulone dissolved in 8 mL of ethanol and 2 mL of pH 10 ammonium hydroxide buffer. The ethanol dissolved the humulone and then adding the ammonium hydroxide immediately changed the color of the sample inside the polarimeter cell. There is little change in the optical rotation degrees (Below).