Materials and Methods
Hops oil is the starting substance to extract the humulone from. o-phenylenediamine is also used alongside benzene. A hot plate, magnetic stir rod, pipette, syringe, Erlenmeyer flask, foil, rubber stop, activated charcoal, Buchner funnel, vacuum filter, filter paper, filter flask, vial, balance, balance paper, rotary evaporator (rotovap), polarimeter, polarimeter cell, round bottom flasks of multiple volumes, bump bulb, magnesium sulfate, hydrochloric acid (6M and 12M), ether, deionized water, pH buffer, deuterated chloroform, NMR tube, separatory funnel, graduated cylinder, and vacuum pump were all used in different quantities throughout the research.
Humulone can be extracted from hops oil. According to F.R. Sharpe and I. H. L. Ormrod, humulone can be extracted and isolated by preparing an o-phenylenediamine/ humulone complex (OPD complex). This is useful to the experiment because humulone needs to be isolated to be able to explore its isomerization rates. With the o-phenylenediamine/ humulone complex, the complex is able to crystallize after being added with benzene as a solvent. This allows for the complex to be more pure after every crystallization.
The complex is crystallized by bringing the complex and benzene to a boil while mixing. The complex is then treated with decolorizing charcoal. Vacuum filtration is next while keeping the complex heated. Once cooling, the complex re-crystallizes with the precipitate left behind. In the lab, this was done two times to be able to extract the humulone.
To re-crystallize, 30.236 g of hops oil, 12.028 g OPD, and 50 mL of benzene were used. The hops oil was heated along with o-phenylenediamine and benzene until the mixture was dissolved and boiling. A Buchner funnel was prepared by placing it in the oven to keep it hot in preparation for filtering. Once the mixture came to a boil, heat was turned off while a pea sized amount of decolorizing charcoal was added and stirred. The mixture was then placed back on the hotplate while the now hot Buchner funnel was placed in the vacuum filtration setup. The mixture was quickly transferred to the vacuum filter and immediately filtered. After filtration, the filter flask was placed in the refrigerator to crystallize for fifteen minutes. After the fifteen minutes, the crystals were removed and vacuum filtered a second time by using benzene. The mass of the remaining yellow crystals was 34.472 g.
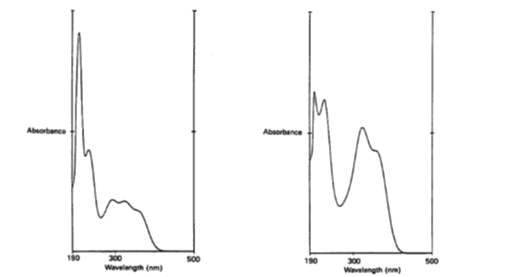
Sharpe and Ormrod also describe how to separate the humulone from the OPD complex. This is the essential part in the research because a supply of humulone is needed to conduct research with. The complex has other compounds, so the purification process will help to get just humulone. Figure 3 shows the difference between purified and non-extracted humulone.
The separation process requires a separatory funnel, the complex, deionized water, hydrochloric acid, ether, magnesium sulfate as a drying agent. 30 mL of 6 M HCl was obtained by mixing 15 mL of DI water and 15 mL of 12 M HCl. 1.05 g of the complex was added with 20 mL ether in a separatory funnel, then shaken and vented. Then 6 mL of 6 M HCl was added to the funnel and shaken and vented. The bottom aqueous layer was separated. The entire process was repeated and then the organic layer was removed. The aqueous layer was re-added to the separatory funnel with ether to collect the aqueous layer again. The organic layer was added back and washed with 6 mL 6M HCl, and then again with about 6 mL brine, and twice more with DI water. The organic layer remaining was put in an Erlenmeyer flask with magnesium sulfate as a drying agent to remove water. The magnesium sulfate was gravity filtered out and the organic layer was put inside of a round bottom flask. The ether was rotovapped out and an orange substance was left over
A proton nuclear magnetic resonance (H-NMR) spectrum was collected from this sample to determine what exactly is in the crystallized humulone. The separation process was repeated with Sean Johnson’s humulone crystals and an H-NMR was also taken of his sample for comparison.
Following US patent 3,354,219, the next goal was to isolate isohumulone. 4 g of hop oil and 25 mL of the pH 10 buffer were refluxed for 10 minutes at a boil. Water was filtered into an Erlenmeyer flask by wetting the filter paper with DI water to separate the water and oil. 4 mL of 6 M HCl was added to the water to crash out the organic material that according to the patent, contains isohumulone. There was no crash out of organic material so the compound was placed in a separatory funnel with ether, shook and vented, and collected the aqueous and ether layers. The ether layer was dried with magnesium sulfate and then filtered into a vial and rotovapped. An H-NMR was taken and the results were inconclusive.
Due to the inconclusive results, the patent was replicated using hops instead of hops oil. 300 mL of DI water, 8.001 g of Australian Galaxy 15% hops, and 10 mL of 3 N NaOH were refluxed in a light-shielded 500 mL round bottom flask for 10 minutes. The mixture was filtered into a 500 mL Erlenmeyer. 7.5 mL of 6 M HCl was added to the water and hops mixture and mixed. The organics with attached isohumulone crashed out. The white organics containing isohumulone was then filtered out of the water under a vacuum filter and Buchner funnel. The organic material and isohumulone left weighed 8.366 g.
The organic material with isohumulone was combined with 15 mL of methanol in attempt to dissolve it. Although not completely dissolved, the solution was placed in a separation funnel with 15 mL of hexanes and 2% sulfuric acid. The funnel was shaken and vented in attempt to dissolve the compound. It dissolved some, so the aqueous layer was going to be drained, but it clogged the separatory funnel. The hexanes layer was needed from the top, so it was pipetted off and filtered. It was then put in a vial and rotovapped. The final compound weight was 0.171 g.
Humulone can be extracted from hops oil. According to F.R. Sharpe and I. H. L. Ormrod, humulone can be extracted and isolated by preparing an o-phenylenediamine/ humulone complex (OPD complex). This is useful to the experiment because humulone needs to be isolated to be able to explore its isomerization rates. With the o-phenylenediamine/ humulone complex, the complex is able to crystallize after being added with benzene as a solvent. This allows for the complex to be more pure after every crystallization.
The complex is crystallized by bringing the complex and benzene to a boil while mixing. The complex is then treated with decolorizing charcoal. Vacuum filtration is next while keeping the complex heated. Once cooling, the complex re-crystallizes with the precipitate left behind. In the lab, this was done two times to be able to extract the humulone.
To re-crystallize, 30.236 g of hops oil, 12.028 g OPD, and 50 mL of benzene were used. The hops oil was heated along with o-phenylenediamine and benzene until the mixture was dissolved and boiling. A Buchner funnel was prepared by placing it in the oven to keep it hot in preparation for filtering. Once the mixture came to a boil, heat was turned off while a pea sized amount of decolorizing charcoal was added and stirred. The mixture was then placed back on the hotplate while the now hot Buchner funnel was placed in the vacuum filtration setup. The mixture was quickly transferred to the vacuum filter and immediately filtered. After filtration, the filter flask was placed in the refrigerator to crystallize for fifteen minutes. After the fifteen minutes, the crystals were removed and vacuum filtered a second time by using benzene. The mass of the remaining yellow crystals was 34.472 g.
Sharpe and Ormrod also describe how to separate the humulone from the OPD complex. This is the essential part in the research because a supply of humulone is needed to conduct research with. The complex has other compounds, so the purification process will help to get just humulone. Figure 3 shows the difference between purified and non-extracted humulone.
The separation process requires a separatory funnel, the complex, deionized water, hydrochloric acid, ether, magnesium sulfate as a drying agent. 30 mL of 6 M HCl was obtained by mixing 15 mL of DI water and 15 mL of 12 M HCl. 1.05 g of the complex was added with 20 mL ether in a separatory funnel, then shaken and vented. Then 6 mL of 6 M HCl was added to the funnel and shaken and vented. The bottom aqueous layer was separated. The entire process was repeated and then the organic layer was removed. The aqueous layer was re-added to the separatory funnel with ether to collect the aqueous layer again. The organic layer was added back and washed with 6 mL 6M HCl, and then again with about 6 mL brine, and twice more with DI water. The organic layer remaining was put in an Erlenmeyer flask with magnesium sulfate as a drying agent to remove water. The magnesium sulfate was gravity filtered out and the organic layer was put inside of a round bottom flask. The ether was rotovapped out and an orange substance was left over
A proton nuclear magnetic resonance (H-NMR) spectrum was collected from this sample to determine what exactly is in the crystallized humulone. The separation process was repeated with Sean Johnson’s humulone crystals and an H-NMR was also taken of his sample for comparison.
Following US patent 3,354,219, the next goal was to isolate isohumulone. 4 g of hop oil and 25 mL of the pH 10 buffer were refluxed for 10 minutes at a boil. Water was filtered into an Erlenmeyer flask by wetting the filter paper with DI water to separate the water and oil. 4 mL of 6 M HCl was added to the water to crash out the organic material that according to the patent, contains isohumulone. There was no crash out of organic material so the compound was placed in a separatory funnel with ether, shook and vented, and collected the aqueous and ether layers. The ether layer was dried with magnesium sulfate and then filtered into a vial and rotovapped. An H-NMR was taken and the results were inconclusive.
Due to the inconclusive results, the patent was replicated using hops instead of hops oil. 300 mL of DI water, 8.001 g of Australian Galaxy 15% hops, and 10 mL of 3 N NaOH were refluxed in a light-shielded 500 mL round bottom flask for 10 minutes. The mixture was filtered into a 500 mL Erlenmeyer. 7.5 mL of 6 M HCl was added to the water and hops mixture and mixed. The organics with attached isohumulone crashed out. The white organics containing isohumulone was then filtered out of the water under a vacuum filter and Buchner funnel. The organic material and isohumulone left weighed 8.366 g.
The organic material with isohumulone was combined with 15 mL of methanol in attempt to dissolve it. Although not completely dissolved, the solution was placed in a separation funnel with 15 mL of hexanes and 2% sulfuric acid. The funnel was shaken and vented in attempt to dissolve the compound. It dissolved some, so the aqueous layer was going to be drained, but it clogged the separatory funnel. The hexanes layer was needed from the top, so it was pipetted off and filtered. It was then put in a vial and rotovapped. The final compound weight was 0.171 g.
UV absorbance spectra by Sharpe and Ormrod. Spectrum of OPD complex (left) and purified humulone (right).